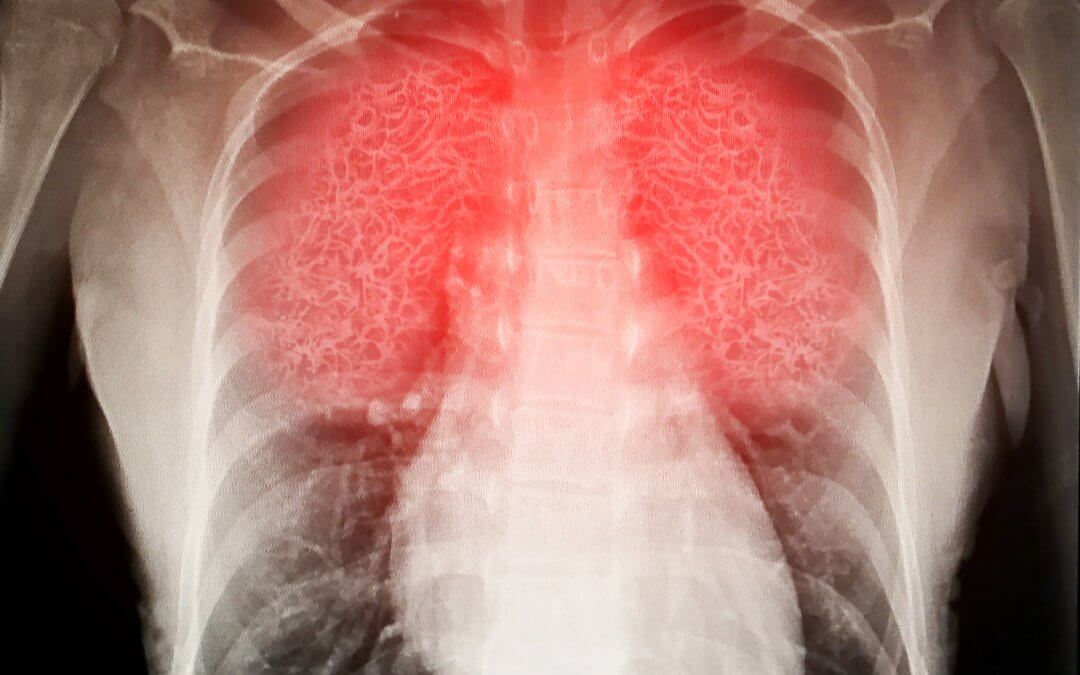
When a severe illness, infection, or injury affects the lungs, inflammation can cause fluid to accumulate, leading to Acute Respiratory Distress Syndrome (ARDS).[1][2] With limited effective treatments, this life-threatening condition remains a major medical challenge. Stem cell therapies are being investigated as a potential supportive treatment; recent reviews summarie current findings and identify areas for further research.[3][4]
What is ARDS?
Normally, small sacs in the lungs called alveoli fill with air during breathing. Oxygen passes from the alveoli into the blood, while carbon dioxide moves from the blood into the alveoli to be exhaled.[5]
In ARDS, inflammation can damage the alveolar membrane, allowing fluid to leak into the alveoli. This reduces oxygen transfer to the blood, which can impair organ function.[1][2] ARDS often develops within hours to days of the illness or injury that triggered it and can worsen rapidly. Complications may include multiple organ failure and cognitive issues due to low oxygen levels.[1][2][6] Mortality in ARDS varies according to severity, with clinical data suggesting rates of approximately 27% in mild cases and up to around 45% in more severe disease.[7] Survivors may experience long-term impacts, including reduced lung function and other consequences related to prolonged hospitalization and mechanical ventilation.[6]
The research so far
A recent review analyzed 48 studies involving 1,773 patients with ARDS treated with mesenchymal stem cells (MSCs) or MSC-derived extracellular vesicles (EVs). Of these, 31 studies (1,321 patients) were suitable for pooled analysis, while the remaining 17 were assessed narratively.
MSCs were derived from sources including bone marrow, adipose tissue, umbilical cord, and placenta. Most studies focused on ARDS related to COVID-19. The review found that MSCs or EVs were generally safe, with no increase in adverse events compared to control groups. Some data suggested a reduction in all-cause mortality within one month, particularly at higher doses, although results were not consistent across all studies.[4]
A second review also confirmed MSC safety even at higher or multiple doses, but highlighted that efficacy appears to depend on the underlying cause of ARDS. For example, COVID-19-related ARDS involves blood vessel lining dysfunction and cytokine storms, where MSCs’ anti-inflammatory effects may improve outcomes.[3][8] Benefits may be less clear for ARDS caused by other triggers such as sepsis, trauma, or pneumonia.[3]
A road to future treatments
Both reviews emphasize that more research is needed. Well-designed, large-scale, double-blinded trials are necessary to determine effective dosages, identify which causes of ARDS respond best, and establish the most suitable source of MSCs.
To learn more about MSCs and other types of stem cells, including current research and potential future applications, fill out the form below to request our free info kit.
References
FIND OUT MORE, REQUEST YOUR FREE INFO KIT TODAY
All you need to know to make an informed decision.
Provide your contact details to request:
– Complete Info Kit and your Guide to Cord Blood Banking
– Information via email
– Contact from our specialist advisors

Lesley-Ann Martin, PhD
Chief Scientific Officer at Cells4Life
Dr. Martin holds a PhD in Molecular Biology from the University of Reading and has extensive research experience at the Institute of Cancer Research. She oversees all scientific and laboratory operations at Cells4Life.