Umbilical cord blood stem cells
Umbilical cord blood stem cells
Umbilical cord blood stem cells
Umbilical cord blood stem cells could change your baby’s life

Powerful

Unique

Safe
Umbilical cord blood is the blood that can be collected from your baby’s umbilical cord and the placenta after they are born. Umbilical cord blood contains billions of powerful stem cells, which are already used to treat over 80 different diseases. They may even hold the key to regenerative medicine. This is why millions of parents in the UK and around the world chose to store blood from their children’s umbilical cord and placenta.
What are stem cells and what can they do?
Stem cells are powerful master cells that have two unique properties:
For example, skin cells act as a protective barrier to the outside world while heart cells pump vital blood around their body. Red blood cells carry the precious oxygen that your child needs to function, and white blood cells form the immune system that prevents them from falling ill.
Given the right conditions and signals, your baby’s umbilical cord blood stem cells can differentiate into these different cell types. As a result, they are extremely valuable in treating an increasing array of medical conditions where specialised cells are damaged and require replacing.
What makes umbilical cord blood stem cells different?
Stem cells are present in many parts of the human body. However, some sources contain richer concentrations than others. Human embryos are also excellent sources of stem cells but their use is controversial and illegal in some countries. In contrast, umbilical cord blood and tissue are collected from material that normally has no use following a child’s birth. They are the richest and purest source of stem cells that your child will ever have.
Storing umbilical cord blood stem cells therefore gives you the opportunity to safeguard your child’s health for years to come. Moreover, they can be easily and safely collected, and reliably stored for treating future health issues. This table compares umbilical cord blood and tissue stem cells with other stem cell sources:
|
|
Cord Blood | Bone Marrow | Peripheral blood | Adipose |
| Potency | ||||
| Availability | ||||
| Routine use in humans | ||||
| Low tumour risk | ||||
| Does not require powerful drugs to collect | ||||
| Contains HSCs | ||||
| Contains MSCs | ||||
| Capacity for high proliferation | ||||
| Low viral contamination risk | ||||
| Non-invasive and painless to collect |
Umbilical cord blood stem cells have the power to repair, regenerate and save lives.
SECURE THIS OPPORTUNITY FOR YOUR UNBORN BABYStem cell types in cord blood
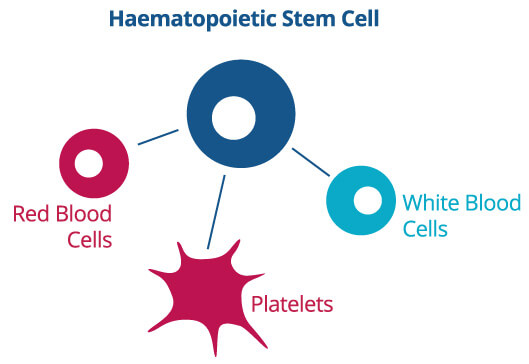
There are three main stem cell types in umbilical cord blood. These are haematopoietic stem cells, mesenchymal stem cells and very small embryonic-like stem cells. Each has its own function:
These cells have the ability to transform into various blood cells, including white blood cells. Medical professionals are using this type of stem cell in many current therapies, such as cancer therapy, to repair the immune system.
<<-Click image to enlarge
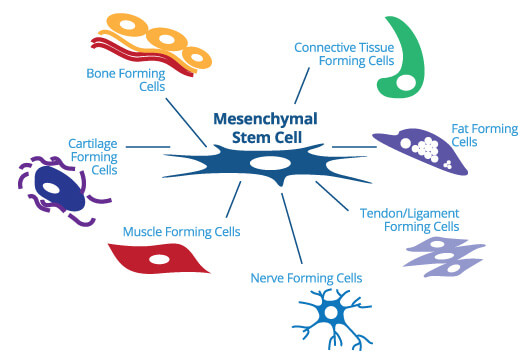
Mesenchymal stem cells (MSCs)
These cells have the ability to transform into a huge range of tissue types, including: nerve tissue / muscle tissue / cartilage.
As a result, these cells are considered extremely important for regenerative therapies. Early studies have shown that mesenchymal stem cells play a crucial role not only in the repair and renewal of these tissue types, but also in regenerating entire organs, such as the heart.
<<-Click image to enlarge
Currently, we know less about the properties of VSELs. However, it is possible that they could be the most valuable stem cell type of all. As their name suggests, VSELs share many of the features of embryonic stem cells. This means they can transform into a wider array of tissue types than most other stem cell types. As a result, the number of potential conditions that they could be used to treat is far higher.
Quick questions…
Can umbilical cord blood be used more than once?
Whether or not you or your family could use their cord blood sample multiple times depends on the service you select and the size of the sample collected.
Our unique CellsPlus processing technology delivers between 2.2 and 3 times more stem cells at the point of therapy than other currently available cord blood processing methods. For this reason, CellsPlus broadens the therapeutic opportunities available to you and your family. Find out more about how CellsPlus compares here.
What is cord stem cell storage?
To ensure quick and efficient cord stem cell storage, Cells4Life processes all umbilical cord stem cells within 24 hours of procurement, with the majority of samples processed in under 12 hours. Samples then undergo cryopreservation, which is a process of freezing the stem cells in the vapour phase of liquid nitrogen until they a required for therapy.
Cells4Life offers multiple sample storage, which involves splitting a sample across two locations. We only have to thaw the required amount of cells for any given therapy, leaving the rest in storage for later use.
Research has shown that stem cells collected at birth can remain viable after storage for at least 25 years and the prevailing view is that it should be possible to store them for much longer this, providing the benefit of umbilical cord blood banking for you and your baby for many years to come.
Are there any ethical issues attached to cord blood stem cells?
What are the advantages of umbilical cord blood stem cells?
Cord Blood Stem Cell Treatment Offers Superior Results for Knee Osteoarthritis, Study Finds
Osteoarthritis, a chronic degenerative joint disease, affects millions of people worldwide and is a leading cause of disability.[1][2] Currently available treatments can offer relief from pain and other symptoms. However, they fail to address the root of the disease,...
Fresh Insights on Cell Therapy for Cerebral Palsy
A new study on cell therapy for cerebral palsy has, for the first time, directly compared the efficacy of two different treatments. Although more research is needed, the study’s results offer valuable insights for future clinical trials tackling this challenging...
Leukaemia Treatment Derived From Cord Blood Gains FDA Approval for Clinical Trials
A CAR-T cell therapy for acute lymphoblastic leukaemia[1] has recently been approved by the FDA for use in clinical trials in the United States. The therapy is based on cells derived from allogeneic (donor) umbilical cord blood; it is the very first such treatment to...
Find out more, request your welcome pack today
Connect with us
* based on our peer reviewed publication showing that TotiCyte delivers 2.2 to 3 times more haematopoietic stem cells at the point of use than other cord blood processing systems in use in the UK.
References
Xiao-Peng Tang et al. “Differentiation of human umbilical cord blood stem cells into hepatocytes in vivo and in vitro” World J Gastronenterology 2006 Jul 7; 12(25):4014-4019 <https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4087712/>
Anna Hordyjewska et al. “Characteristics of hematopoietic stem cells of umbilical cord blood” Cytotechnology 2015 May; 67(3):387-396 <https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4371573/>
Ratajczak MZ, et al. “Umbilical cord blood-derived very small embryonic like stem cells (VSELs) as a source of pluripotent stem cells for regenerative medicine.” Pediatr Endocrinol Rev. 2012 Mar;9(3):639-43 <https://www.ncbi.nlm.nih.gov/pubmed/22523831>
Benedetta Bussolati, “Stem cells for organ repair” Organogenesis 2011 Apr-Jun; 7(2): 95 <https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3142444/>
Levitt, Alice. “A Ghost (Heart) of a Chance” Houstonia Magazine, accessed 18 March 2019. <https://www.houstoniamag.com/articles/2017/1/23/texas-heart-institute-new-research-ghost-heart>
N.I. Kalinina et al. (2011) “Mesenchymal Stem Cells in Tissue Growth and Repair” Acta Nature, <https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3347612/>
Miao et al. (2017) “A brief review: the therapeutic potential of bone marrow mesenchymal stem cells in myocardial infarction”, Stem Cell Research & Therapy <https://stemcellres.biomedcentral.com/articles/10.1186/s13287-017-0697-9>
Nguyen, L.T., Tran, N.T., Than, U.T.T. et al. Optimization of human umbilical cord blood-derived mesenchymal stem cell isolation and culture methods in serum- and xeno-free conditions. Stem Cell Res Ther 13, 15 (2022). https://doi.org/10.1186/s13287-021-02694-y
Urrutia DN, Caviedes P, Mardones R, Minguell JJ, Vega-Letter AM, et al. Comparative study of the neural differentiation capacity of mesenchymal stromal cells from different tissue sources: An approach for their use in neural regeneration therapies. PLOS ONE. 2019; 14(3): e0213032. https://doi.org/10.1371/journal.pone.0213032
Choi, Y. S., Dusting, G. J., Stubbs, S., Arunothayaraj, S., Han, X. L., Collas, P., Morrison, W. A., & Dilley, R. J. (2010). Differentiation of human adipose-derived stem cells into beating cardiomyocytes. Journal of cellular and molecular medicine, 14(4), 878–889. https://doi.org/10.1111/j.1582-4934.2010.01009.x
Hang H., Yu Y., Wu N., Huang Q., Xia Q., Bian J. Induction of highly functional hepatocytes from human umbilical cord mesenchymal stem cells by HNF4alpha transduction. PLoS One. 2014;9:e104133. doi: 10.1371/journal.pone.0104133
Prabakar K.R., Dominguez-Bendala J., Molano R.D., Pileggi A., Villate S., Ricordi C., Inverardi L. Generation of glucose-responsive, insulin-producing cells from human umbilical cord blood-derived mesenchymal stem cells. Cell Transplant. 2012;21:1321–1339. doi: 10.3727/096368911X612530.
Zhang X., Hirai M., Cantero S., Ciubotariu R., Dobrila L., Hirsh A., Igura K., Satoh H., Yokomi I., Nishimura T., et al. Isolation and characterization of mesenchymal stem cells from human umbilical cord blood: reevaluation of critical factors for successful isolation and high ability to proliferate and differentiate to chondrocytes as compared to mesenchymal stem cells from bone marrow and adipose tissue. J. Cell. Biochem. 2011;112:1206–1218. doi: 10.1002/jcb.23042
Heris, R.M., Shirvaliloo, M., Abbaspour-Aghdam, S. et al. The potential use of mesenchymal stem cells and their exosomes in Parkinson’s disease treatment.Stem Cell Res Ther 13, 371 (2022). https://doi.org/10.1186/s13287-022-03050-4
Li, Z., Dong, X., Tian, M. et al. Stem cell-based therapies for ischemic stroke: a systematic review and meta-analysis of clinical trials. Stem Cell Res Ther 11, 252 (2020). https://doi.org/10.1186/s13287-020-01762-z
Karvelas N, Bennett S, Politis G, Kouris NI, Kole C. Advances in stem cell therapy in Alzheimer’s disease: a comprehensive clinical trial review. Stem Cell Investig. 2022 Feb 21;9:2. doi: 10.21037/sci-2021-063. PMID: 35280344; PMCID: PMC8898169.
Choudhury, T., Mozid, A., Hamshere, S., Yeo, C., Pellaton, C., Arnous, S., Saunders, N., Brookman, P., Jain, A., Locca, D., Archbold, A., Knight, C., Wragg, A., Davies, C., Mills, P., Parmar, M., Rothman, M., Choudry, F., Jones, D. A., Agrawal, S., … Mathur, A. (2017). An exploratory randomized control study of combination cytokine and adult autologous bone marrow progenitor cell administration in patients with ischaemic cardiomyopathy: the REGENERATE-IHD clinical trial. European journal of heart failure, 19(1), 138–147. https://doi.org/10.1002/ejhf.676
Jiang, Y., Jahagirdar, B. N., Reinhardt, R. L., Schwartz, R. E., Keene, C. D., Ortiz-Gonzalez, X. R., Reyes, M., Lenvik, T., Lund, T., Blackstad, M., Du, J., Aldrich, S., Lisberg, A., Low, W. C., Largaespada, D. A., & Verfaillie, C. M. (2002). Pluripotency of mesenchymal stem cells derived from adult marrow. Nature, 418(6893), 41–49. https://doi.org/10.1038/nature00870
Zhou, M., Xi, J., Cheng, Y. et al. Reprogrammed mesenchymal stem cells derived from iPSCs promote bone repair in steroid-associated osteonecrosis of the femoral head. Stem Cell Res Ther 12, 175 (2021). https://doi.org/10.1186/s13287-021-02249-1