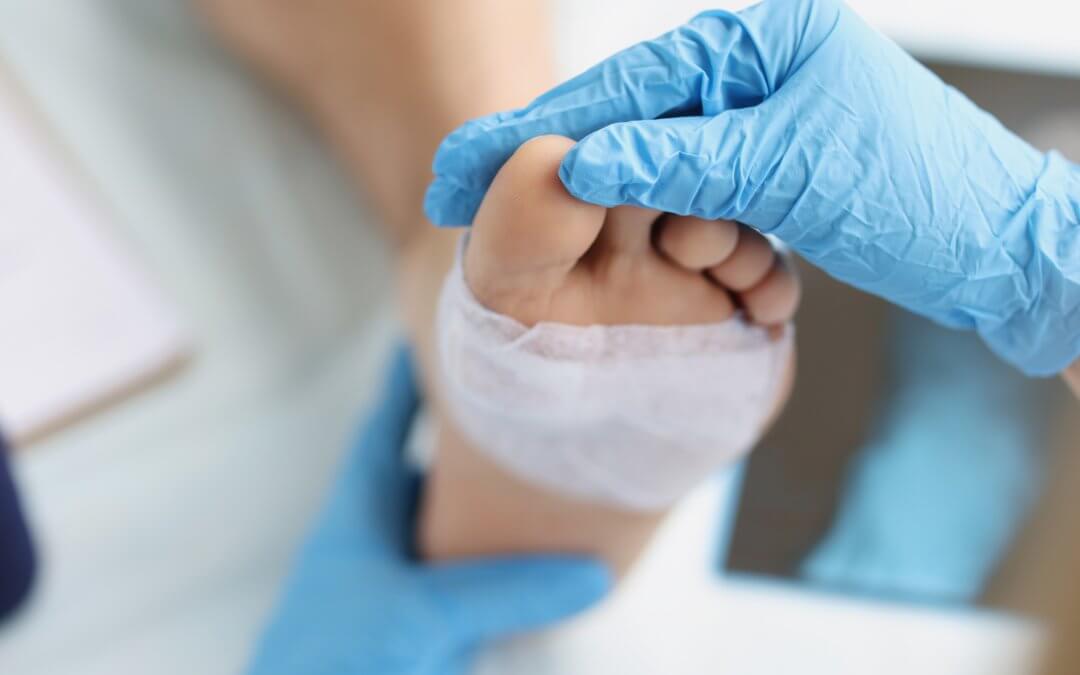
Key details
- High amputation risk: Diabetic foot ulcers are a serious complication of diabetes, often leading to infection and significantly higher amputation risks compared to the non-diabetic population.[1]
- Treating the root cause: Stem cell therapies, particularly mesenchymal stem cells, are being investigated for their potential to support wound healing by promoting blood vessel formation, modulating inflammation, and influencing immune responses. These effects may help improve the local healing environment in diabetic foot ulcers.[2]
- Source matters: While bone marrow and adipose (fat) tissue are established sources of stem cells, umbilical cord–derived mesenchymal stem cells (UC MSCs) are also widely studied because they can be collected non-invasively from tissue normally discarded at birth, such as umbilical cord tissue or cord blood. These neonatal cells have demonstrated a high capacity for proliferation and expansion in laboratory studies, making them a promising resource for research into future regenerative therapies. Clinical studies are ongoing to determine their safety, efficacy, and potential applications.[3][4]
What are diabetic foot ulcers and why are they dangerous?
Diabetic foot ulcers are chronic wounds caused by a combination of nerve damage (neuropathy) and restricted blood supply, which can lead to severe infection and tissue death.
Because diabetic neuropathy causes loss of sensation in the legs and feet, small nicks and wounds frequently go unnoticed and untreated, leading them to ulcerate or become infected. Simultaneously, damage to blood vessels restricts the blood supply, meaning the feet receive fewer infection-fighting cells, causing wounds to heal slowly and potentially become gangrenous.[5][6]
The statistics regarding this condition:
- Prevalence: Up to one third of people with diabetes will develop a foot ulcer during their lifetime.[7]
- Amputation risk: The risk of leg, foot, or toe amputation for people with diabetes is more than 20 times that of people without diabetes. Up to 28% of diabetic foot ulcers could result in some form of amputation.[8]
- Mortality: People who develop diabetic foot ulcers or require amputation often have other serious health conditions, and overall survival can be reduced. Studies suggest that around 35–50% of patients may die within five years of developing a foot ulcer, with higher risks in those who require amputation. Outcomes vary depending on overall health, ulcer severity, and access to care.[8]
How are diabetic foot ulcers currently treated?
Current treatment for diabetic foot ulcers focuses mainly on symptom management, involving wound care, offloading pressure, and infection control.
Standard protocols include dressings, antibiotics, compression clothes, and keeping weight off the foot using orthotics, special footwear, or wheelchairs. More invasive treatment, including debridement (surgical removal of infected tissue), may also be necessary.[9]
However, these therapies often fail to achieve complete healing. Only two thirds of diabetic foot ulcers eventually heal without surgery.[5][7][8]
How could stem cell therapy help heal diabetic ulcers?
Therapies based on mesenchymal stem cells (MSCs) have emerged as a promising strategy for addressing the underlying pathophysiology of diabetic foot ulcers (DFUs). MSCs exert multifactorial effects, including immunomodulation, anti-inflammatory activity, angiogenic promotion, and recruitment of reparative immune cells to the site of injury. These properties collectively target the principal contributors to impaired healing in DFUs, namely, chronic inflammation, abnormal immune responses, and inadequate microvascular perfusion, mechanisms that conventional therapies often fail to correct.[2][10]
Preclinical and clinical investigations have explored the therapeutic potential of MSCs in DFUs, demonstrating encouraging outcomes. A 2022 meta-analysis, aggregating data from 14 studies, reported that stem cell therapies significantly outperformed standard care across several critical endpoints:[11]
- Enhanced wound healing: Higher rates of ulcer closure and improved local blood flow.
- Pain amelioration: Increased pain-free walking distance and reduced pain at rest.
- Limb preservation: A notable reduction in lower-limb amputation rates.
These findings suggest that MSC-based therapies offer a mechanistically rational and clinically beneficial approach to DFU management, addressing not only symptomatic healing but also the underlying biological deficiencies that drive chronic, non-healing wounds.[2][10][11]
What challenges remain for widespread clinical use?
Despite encouraging results, challenges such as patient variability and the need for standardised data prevent the treatment from being ready for widespread use immediately.
Small differences between patients, their age, how well their diabetes is controlled, vascular disease, and the microorganisms inhabiting the ulcer, can have an impact on how the treatment works. This makes it complicated to assemble a single set of coherent, standardised data from multiple small-scale trials. Thus, larger-scale trials with a lengthy follow-up period will be required to ensure the treatment is effective and remains so long-term.[12]
Which types of stem cells could be effective for treating diabetic foot ulcers?
Research currently focuses on Mesenchymal Stem Cells (MSCs) derived from bone marrow, adipose (fat) tissue, or umbilical cord blood, though each source presents different advantages and challenges.
- Bone Marrow MSCs: They have an established clinical track record and show strong potential for vascular regeneration. However, they require invasive collection procedures. Furthermore, their regenerative potential may be impaired by the donor’s age, as well as, in the case of autologous (using the patient’s own cells) therapies, by abnormal bone marrow microenvironment caused by diabetes.
- Adipose (Fat) MSCs: They are easy to collect and expand in vitro. Research has found that they can promote wound healing, reduce inflammation, and improve tissue regeneration. However, their function may be compromised by the patient’s weight and diabetes-related lipotoxicity (fat accumulation in organs) can also reduce these cells’ ability to migrate to the wound site.
- Umbilical Cord MSCs: These cells have high self-renewal abilities, strong immunoregulatory functions, and can promote angiogenesis and reduce infection risk.[12] However, access to them is limited by the fact that they can only be collected immediately after birth.
How can parents prepare for the potential of future stem cell treatments?
Research is ongoing into the use of stem cells, including those from umbilical cord blood, for a variety of conditions, such as diabetic complications. If parents choose to collect and store their baby’s cord blood at birth, these cells could be available for future research or potential treatments.
For parents who want to learn more about cord blood collection and storage, our free guide explains the process and options.
References
FIND OUT MORE, REQUEST YOUR WELCOME PACK TODAY
All you need to know to make an informed decision.
Provide your contact details to request:
– Complete Welcome Pack and Parent’s Guide
– Information via email
– Contact from our specialist advisors

Lesley-Ann Martin, PhD
Chief Scientific Officer at Cells4Life
Dr. Martin holds a PhD in Molecular Biology from the University of Reading and has extensive research experience at the Institute of Cancer Research. She oversees all scientific and laboratory operations at Cells4Life.