Key details
- Disabling condition: Duchenne muscular dystrophy (DMD) is a severe, genetic condition that causes progressive muscle weakness due to the absence of dystrophin, a protein essential for maintaining muscle integrity.
- Increasing scientific interest in whether stem cell-based therapies could play a role in treating Duchenne muscular dystrophy (DMD). While this research is still at an early stage, recent updates from the U.S. Food and Drug Administration (FDA) highlight ongoing efforts to explore these approaches.
- EN001 orphan drug designation: The U.S. Food and Drug Administration (FDA) has granted orphan drug designation to EN001, an investigational therapy derived from umbilical cord mesenchymal stem cells for Duchenne muscular dystrophy (DMD). This designation is intended to support research into the development of treatments for rare diseases by. It is important to note that this status does not mean the therapy has been proven to be safe or effective.
- Ryoncil clinical trial authorisation: Ryoncil (remestemcel-L), an investigational stem cell therapy derived from bone marrow, has been authorised by the FDA to be studied in a clinical trial for DMD. This allows researchers to assess its safety and potential effectiveness in patients. If the trial shows positive results, the findings may support a future application for regulatory approval. However, the therapy is not currently approved for the treatment of DMD and remains under investigation.
What is Duchenne muscular dystrophy?
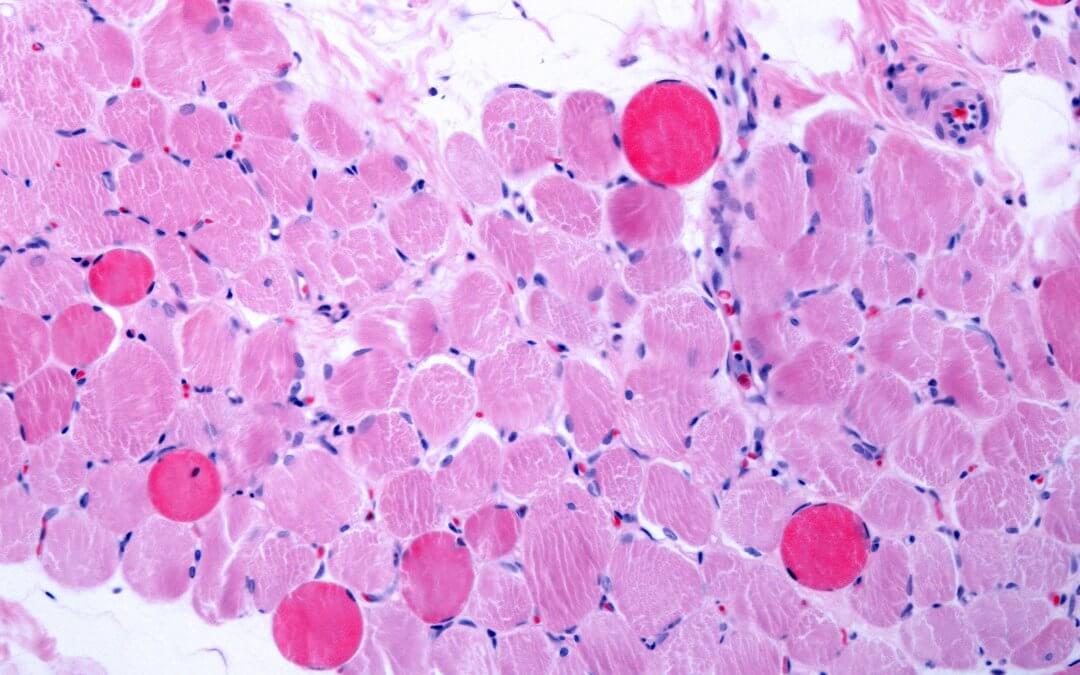
Duchenne muscular dystrophy (DMD) is a serious inherited condition that causes muscles to become weaker over time. It happens because of a change (mutation) in the gene that makes dystrophin, a protein that helps keep muscle cells strong and protected. Without dystrophin, muscle cells are easily damaged and gradually break down. Over time, the body cannot properly repair this damage, and muscle tissue is replaced by fat and scar tissue [1][2].
DMD is linked to the X chromosome, which means it mainly affects boys. It occurs in around 1 in every 3,500–5,000 male births [3]. Girls can carry the gene but usually have few or no symptoms because they have a second X chromosome that can compensate. However, they have a 50% chance of passing the condition on to their sons. In about 30% of cases, DMD occurs without any family history, due to a new genetic change [2].
Signs of DMD usually appear in early childhood, often between the ages of 2 and 4. Children may have trouble running, jumping, or climbing stairs, find it difficult to stand up from the floor, fall more often than other children, or walk on their toes or with a waddling movement [1][2][3]. The condition gets worse over time, and most boys lose the ability to walk by around age 11 or 12. As it progresses, DMD also affects the heart and breathing muscles, which can lead to serious health problems. With advances in care, many people with DMD are now living into early adulthood [1][2].
There is currently no cure for DMD. Treatment focuses on managing symptoms, maintaining mobility for as long as possible, and slowing down the progression of the condition [1][2][3].
Stem cell therapies in development
Stem cell therapies for DMD are still in the development and clinical trial stages, and as such are not widely available to patients. Still, recent updates in the field offer hope that therapies based on mesenchymal stem cells (MSCs) may be able to alter the course of the disease.
It was recently announced that the U.S. Food and Drug Administration (FDA) granted orphan drug designation to EN001, an investigational umbilical cord MSC therapy, for DMD. This qualifies the therapy for incentives aimed at supporting the development of treatments for rare diseases; the same therapy has also been granted orphan drug designation for Charcot-Marie-Tooth disease, a rare inherited condition that also results in movement difficulties, although it is not as severe as DMD[4].
Results published from a small, phase 1 study of EN001 indicate that the therapy is safe, with no serious treatment-related adverse events reported over the 12-week follow-up period. Although this was mainly a safety study, exploratory evaluation of effectiveness showed potential stabilisation of the disease. Patients showed maintained or improved lung capacity, stable leg strength, and stable levels of creatine kinase (CK), an enzyme which is present in the blood at elevated levels when muscles break down [5]. Following phase 1, an expanded phase 2 trial in South Korea was also planned, although no updates or results have been published yet [6].
Another recent FDA announcement concerns Ryoncil (remestemcel-L-rknd), an investigational bone marrow MSC therapy. This therapy, already available in the US for the treatment of steroid-resistant acute graft-versus-host disease in children, has now been approved for evaluation in a clinical trial for DMD. The trial aims to enrol 76 children with DMD who are receiving standard-of-care treatment; participants will be randomly assigned to receive either the therapy or a placebo. The main goal is to determine whether the therapy improves performance on a time-to-stand test, which is a standard measurement of motor function. Because the trial is designed as a registrational trial, positive results could be used as a basis to apply for FDA approval of the therapy [7].
Notably, Ryoncil and EN001 represent a different therapeutic approach to DMD, focusing on controlling inflammation and immune activity rather than directly addressing the underlying genetic mutation. If the upcoming trial is successful, it could open the door to a new treatment option that helps slow disease progression and improve quality of life for many children.
As regenerative medicine continues to advance, stem cell therapies are being actively researched for a wide range of medical conditions. While many of these applications are still in development and not yet established treatments, the field is progressing and may support further clinical research over time.
If you are expecting, storing your baby’s stem cells is an option some families consider as part of their birth planning. It allows the preservation of stem cells for potential future research or medical use, although the likelihood and nature of any future applications remain uncertain.
To find out more about stem cell banking and how it works, complete the form below to receive your free guide.
References
FIND OUT MORE, REQUEST YOUR WELCOME PACK TODAY
All you need to know to make an informed decision.
Provide your contact details to request:
– Complete Welcome Pack and Parent’s Guide
– Information via email
– Contact from our specialist advisors

Lesley-Ann Martin, PhD
Chief Scientific Officer at Cells4Life
Dr. Martin holds a PhD in Molecular Biology from the University of Reading and has extensive research experience at the Institute of Cancer Research. She oversees all scientific and laboratory operations at Cells4Life.